Personal Care

Product Categories
White Label vs Customization
Want to launch fast or stand out with small changes?
Want to create something entirely new?
MOQs by Product Type
Lead Times by Product Type
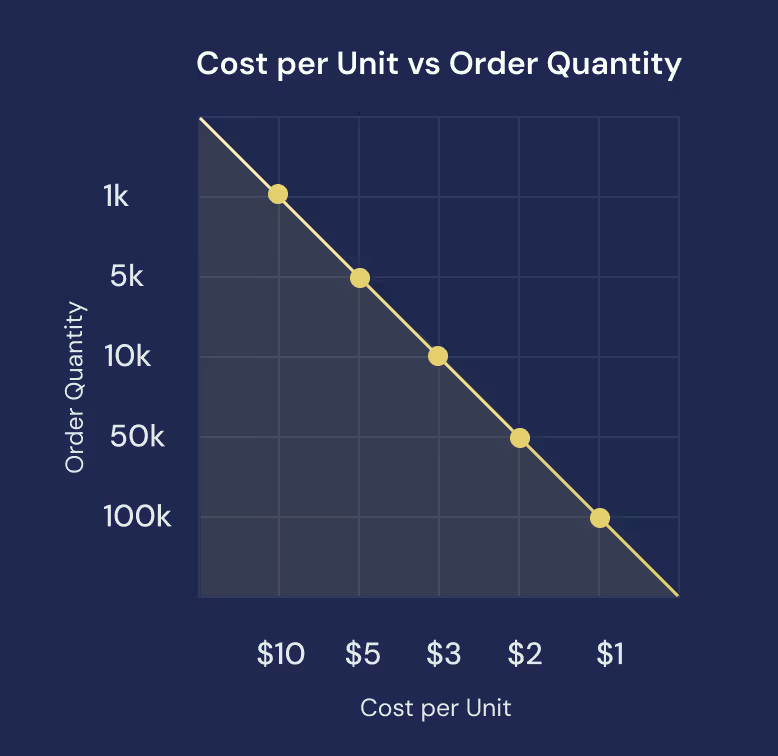
Cost Levers & Unit Economics
Best Manufacturing Countries & When to Choose Each

China leads global cosmetic manufacturing with ISO 22716-certified facilities, extensive ingredient supply chains (including K-beauty and J-beauty actives), and the capacity for both boutique formulations and mass production. Ideal for custom development, cost-effective scaling, and access to specialized cosmetic manufacturers.
- ISO 22716 (Cosmetic GMP) certified facilities widely available
- Custom formulation development with cosmetic chemists
- Among the most cost-effective per-unit pricing at scale (5,000+ units)
- Complete ingredient supply chain (actives, botanicals, preservatives)
- Flexible MOQs (3,000-5,000 pcs depending on complexity)
- Advanced packaging options (airless, droppers, sustainable materials)
- 4-8 week timelines for white label, 10-16 weeks for custom

U.S. cosmetic manufacturing offers simplified FDA compliance, faster communication cycles, shorter shipping times, and easier ingredient verification. Best for brands prioritizing "Made in USA" positioning, rapid iteration, clean beauty with domestic ingredient sourcing, or products requiring frequent reformulation.
- Simplified FDA cosmetic registration and labeling
- Faster sampling cycles (3-5 days vs 2-3 weeks)
- Direct access to clean beauty ingredient suppliers
- No import duties or customs delays
- "Made in USA" brand positioning for marketing
- Smaller test runs possible (1,000-2,500 units)
- 3-6 week production timelines

South Korea specializes in cutting-edge skincare formulations, innovative delivery systems (cushion compacts, ampoules), and trend-forward beauty products. Best for brands wanting access to K-beauty innovation, fermented ingredients, or products that command premium pricing through Korean beauty positioning.
- Access to latest K-beauty trends and formulations
- Fermented ingredients and innovative actives
- Advanced delivery systems (cushions, essences, ampoules)
- Strong regulatory framework and testing standards
- Premium brand positioning through Korean origin
- MOQs typically higher (5,000-10,000 units)
- 8-12 week production timelines
From Brief to First Ship
White Label Fast-Track
Select Formula & Review Samples
Choose from our catalog of pre-developed health and wellness
productChoose from our catalog of pre-developed beauty formulations—face creams, serums, cleansers, haircare, or color cosmetics. We send samples from ISO 22716-certified manufacturers to your location. Test texture, performance, and scent to verify the formula meets your brand standards before committing to customization.
- Texture and absorption testing
- Fragrance profile evaluation
- Packaging format assessment

Brand Customization & Packaging Design
Once you approve the formula, we apply your brand identity through custom labels, packaging design, and visual elements. Our team coordinates with the factory to ensure cosmetic compliance on labels (ingredients in INCI format, proper claims language). You review packaging mockups and label artwork before production..
- INCI ingredient listing and compliance review
- Custom label design and brand application
- Packaging mockup approval

Production, Quality Control & Shipment
Factory begins production with your custom branding applied. Our team conducts on-site quality inspections to verify batch consistency, packaging integrity, and cosmetic compliance. After final approval, we arrange freight, prepare customs documentation including cosmetic declarations, and coordinate delivery to your warehouse or fulfillment center..
- Batch consistency verification
- Fill weight and packaging QC
- Cosmetic declaration and customs coordination

Customization
Formula Development & Ingredient Selection
Work directly with cosmetic chemists to develop your custom beauty formulation. Define active ingredient concentrations (retinol, vitamin C, peptides), select botanical extracts, specify texture and viscosity, choose fragrance profiles, and establish preservative systems. Factory creates initial samples for your evaluation.
- Custom ingredient profile and concentrations
- Texture and viscosity specifications
- Initial formula samples produced

Stability Testing & Formula Refinement
Review initial samples and provide feedback on performance, texture, scent, or efficacy. Factory adjusts formulation based on input. Conduct stability testing (heat/cold cycling, preservative challenge testing) to verify shelf life and product safety. Additional samples produced for final approval.
- Formula refinement based on feedback
- Stability and preservative challenge testing
- Compatibility testing with packaging

Packaging Development & Regulatory Prep
Once formula is approved, finalize packaging specifications—select bottles, jars, pumps, or airless containers. Create custom molds if needed for unique packaging shapes. Compile regulatory documentation (stability data, safety assessments, ingredient disclosures). Finalize label artwork with proper cosmetic compliance.
- Custom packaging selection and mold creation
- Cosmetic safety assessment and documentation
- FDA-compliant label design approva

Production, Batch Testing & Delivery
Full-scale production begins with your custom formulation and packaging. Our Shenzhen team monitors batch consistency, conducts fill weight verification, and ensures cosmetic GMP compliance throughout manufacturing. Final pre-shipment inspection completed before arranging freight and customs clearance.
- Production with batch consistency monitoring
- Cosmetic GMP compliance verification
- Pre-shipment inspection and freight coordination

Ready to Launch Your Beauty Brand?
Checklists
Cosmetic Compliance Requirements
- ISO 22716 (Cosmetic GMP) – manufacturing under Good Manufacturing Practices for cosmetics
- INCI Ingredient Listing – proper International Nomenclature Cosmetic Ingredient format
- Stability Testing Data – heat/cold cycling and shelf life verification
- Safety Assessment (CPSR for EU) – cosmetic product safety report if selling in Europe
- Preservative Challenge Testing – microbiological safety verification
- Allergen Declaration – proper labeling of known allergens
Quality Control Standards
- Batch Consistency Testing – verify each production run matches approved formula
- Fill Weight Verification – ensure accurate product volumes
- pH Testing – critical for skincare stability and skin compatibility
- Viscosity Testing – texture consistency across batches
- Color Matching (for cosmetics) – shade consistency verification
- Packaging Integrity – leak testing and component compatibility
What Can Go Wrong
Factory
What can go wrong:
- Inconsistent batch colors or textures due to raw material variations
- Separation or instability in formulations (oil/water separation, precipitation)
- Contamination during filling process affecting product safety
- Incorrect preservative levels leading to microbial growth
How we prevent it:
- Raw material lot testing before production begins
- In-process quality checks during filling and packaging
- Preservative challenge testing to verify antimicrobial efficacy
- Batch retention samples for traceability
Trade Company
What can go wrong:
- Cannot verify actual factory ISO 22716 certification
- Ingredient sourcing hidden, risk of contaminated or fake actives
- No access to stability testing or safety documentation
- Formula changes between batches without notification
How we prevent it:
- Work directly with ISO 22716-certified factories, no middlemen
- Verify ingredient COAs (Certificates of Analysis) from suppliers
- Maintain full chain-of-custody documentation
- Regular factory audits and batch monitoring
Fulfillment Company
What can go wrong:
- Temperature-sensitive products (vitamin C, retinol) degrading in storage
- Exposure to light breaking down active ingredients
- Improper handling causing leaks or contamination
- Mixing up different SKUs or production batches
How we prevent it:
- Climate-controlled warehousing for temperature-sensitive products
- Light-protected storage for photosensitive actives
- Barcode-based lot tracking system
- First-Expired-First-Out (FEFO) inventory management
Get Your Free Beauty Product Sourcing Checklist
Get Connected
As a loose guide, the typical “sweet spot” Kanary client who will benefit the most from our process and scaleable expertise will have annual gross revenue starting at approximately 5 million USD. If your business meets—or is approaching—this threshold, we can help you!









.avif)
